




The "Bioactive Molecules" team, thanks to the different groups constituting it, focuses on the synthesis of molecules with antiviral or antitumor properties,
as well as biological tools (inhibitors, fluorescent probes, diagnostic tests, etc.) needed to the study of their activity in vitro. The team is also developing
new methods of immunoassays for the pharmacological study of drugs used in antiviral therapy, and multifunctional nanocarriers used in particular to improve the
transfer of genes (gene therapy) and / or cell therapy.
The team (CNRS Website) is working especially on oriented thematic such as design and implementation of nucleosides analogues
and oligonucleotides as therapeutic and biological tools. An important activity is the development of tools to study nucleic acid / protein interactions. Our approach
is interdisciplinary: the chemistry of biological processes is studied by physical methods (spectroscopy) based on the skills of organic chemistry and biochemistry
present in the team. One of the major challenges of the post-genomic era and proteomics concerns the characterization and quantification of protein / nucleic acid
interactions. In addition to the fundamental aspects of these studies, they also aim to understand the molecular basis of various diseases and to develop diagnostic
methods and high throughput screening. To achieve these goals, we must nevertheless have nucleobases sensitive to environmental changes that disrupt as low as possible
interfere with the structure and function of these nucleic acids. In this context we explored to access of new tools based on different approaches to label nucleic
acids and in particular in the development of new fluorescent probes.
If you want to join us, click on the 'Career tab' to submit your application.
Nucleic acids and the development of new tools are the main objectives of our research activities. We introduced a new concept based on the fluorescent probes properties
to emit at two different wavelengths for an internal and external labeling of nucleic acids. These tools are able to translate with high sensitivity nucleic acid /
protein interactions. This project was supported by an 'ANR Blanche' (2007-2011). It is part of a collaboration with Pr. Yves Mely (  , - Faculty of Pharmacy, Strasbourg) for the photophysical
studies and interaction with proteins, Pr. Alexander Demchenko (Palladin Institute of Biochemistry, Kiev) and Professor Vasyl Pivovarenko (
, - Faculty of Pharmacy, Strasbourg) for the photophysical
studies and interaction with proteins, Pr. Alexander Demchenko (Palladin Institute of Biochemistry, Kiev) and Professor Vasyl Pivovarenko (  - Kiev National Taras Shevchenko,
chemistry Faculty) for their expertise in the chemistry of fluorescence probes, and Dr. Cyril Kenfack (
- Kiev National Taras Shevchenko,
chemistry Faculty) for their expertise in the chemistry of fluorescence probes, and Dr. Cyril Kenfack (  - University of Douala) for his contributions to modeling studies and
ab initio calculations.
- University of Douala) for his contributions to modeling studies and
ab initio calculations.
Our expertise in this field has enabled us to propose these photophysical tools to study different biological processes involving nucleic acid / protein interactions.
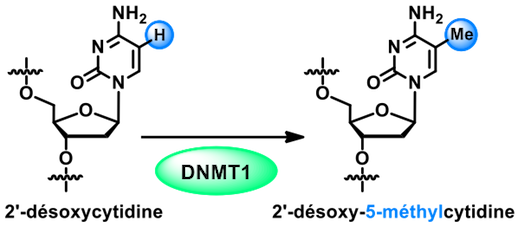
 , - Faculty of Pharmacy, Strasbourg). The study of the 1st step of this epigenetic mechanism (the recognition of hemi-methylated double strand by the SRA domain of UHRF1) was concluded by a publication in the excellent J. Am. Chem. Soc. chemistry journal in 2017.
, - Faculty of Pharmacy, Strasbourg). The study of the 1st step of this epigenetic mechanism (the recognition of hemi-methylated double strand by the SRA domain of UHRF1) was concluded by a publication in the excellent J. Am. Chem. Soc. chemistry journal in 2017.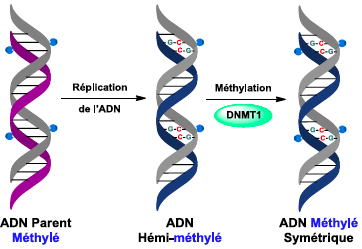
Another project was initiated in collaboration with Pr. Olga Fedorova (  - Novosibirsk State University) who is interested in studying DNA repair mechanisms (Nobel Prize in chemistry 2015). A better
understanding of these mechanisms is an important issue for the Life Sciences and human health. This project is supported by the PACA region through an exploratory project
(2014-2015) and was concluded by three publications (PlosOne 2014, Russ. J. Bioorg. Chem. 2019 (1), Russ. J. Bioorg. Chem. 2019 (2)). We were also interested in HIV Reverse Transcription (Chem. Eur. J. 2018). Finally, we also studied lipid order in biomembranes (ACS Chem. Biol. 2017).
- Novosibirsk State University) who is interested in studying DNA repair mechanisms (Nobel Prize in chemistry 2015). A better
understanding of these mechanisms is an important issue for the Life Sciences and human health. This project is supported by the PACA region through an exploratory project
(2014-2015) and was concluded by three publications (PlosOne 2014, Russ. J. Bioorg. Chem. 2019 (1), Russ. J. Bioorg. Chem. 2019 (2)). We were also interested in HIV Reverse Transcription (Chem. Eur. J. 2018). Finally, we also studied lipid order in biomembranes (ACS Chem. Biol. 2017).
Studying such biological mechanisms is possible thanks to the extreme sensitivity of our fluorescent reporters to environmental changes (Chem. Eur. J. 2014, Chem. Eur. J. 2016), but more particularly hydration (RSC Adv. 2015, Dyes Pigm. 2019). Incorporation of these fluorescently labelled nucleosides into DNA oligonucleotides (ODNs) also allowed us to follow DNA conformational changes, commonly encountered in nucleo-proteic interactions; from single strand to B DNA and/or A DNA (J. Mater. Chem. C 2015) as well as mismatching (RSC Adv. 2016). Förster Resonnance Energy Tranfser (FRET) is also a must have to follow biological interactions. That is the reason why we are also working on the development of highly photostable fluorescent donors presenting a Mega-Stokes Shift [>100 nm] (J. Org. Chem. 2016).